Ca element configuration8/19/2023  varies by element, we have to look at each element on a case-by-case basis to see how these will fill. In the d block it is the other way around.īecause the difference in energy levels between 4s-3d, 5s-4d, etc. The explanation is simple: in Groups 1 and 2, but not in the d block, the s is lower in energy than the d. there is the d to take into consideration. For the first few members to of Groups 1 and 2 there is no d to be filled. This leaves us with why you have s filling in Groups 1 and 2 instead of the d. And in palladium alone, the difference between d and s is so great that none of the electron are in the s. Sometimes the difference in energy levels between the s and d allows one electron to enter the s, sometimes it the difference allows 2 electrons to enter the s. The same process happens throughout the d-block. The difference in energy levels between 4s and 3d is small enough for the next two electrons to enter the 4s, because the electron-electron repulsion they feel if they enter 3d is enough to knock them into the 4s. So, with scandium, the first electron after completing the previous noble gas configuration goes into 3d. So, we have 3d and 4s co-filling rather than filling independently. However, because the 3d orbitals are smaller and more closely packed than the 4s, the electron-electron repulsion that occurs in the 3d subshell is enough to make it possible for 1 or 2 electrons to have a lower energy by occupying the 4s. In scandium and the other d block members of period 4, the 3d is lower in energy than the 4s. How much of a difference, and which subshell is lower in energy, varies by element. The 4s and 3d subshells have nearly the same energy level. I have assumed that the configuration that follows the rules is the ground state, since that is what is usually taught in general chemistry. Note: Nickel has two electron configurations that are commonly encountered over which there is a dispute amongst chemists as to which should be considered the ground state. There is a simple chart that makes learning the filling order easy. He did not know that La is an exception to the rule. Note: in one of his videos, Sal does the configuration of La, but he gets it wrong. However, it is unlikely you would be asked to know these in a general chemistry course. Thus, the actual configuration of the Pd is 4d¹⁰ (the 5s is empty).Īll the other exceptions are in the f block and just have to be learned on a case-by-case basis. Both of these electrons are instead in the outermost d subshell. In Pd (palladium) the outermost s subshell, predicted to have 2 electrons, is actually empty. In the following elements, there is 1 electron, not the predicted 2, in the outermost s subshell, the "missing" electron is instead located in the outermost d subshell: So, rather than going through the far more difficult to understand rules that do correctly predict the electron configurations for all elements, it is easier just to learn the elements that this simplified method doesn't work correctly for. 
There are similar irregularities in the filling of 5s and 4d as well as some other pairs of subshells. For example, the 4s and 3d have nearly the same energy level and so, in a way that varies according to which element it is, either of the two subshells could be the first to fill and there is no certainty that one will be completely filled before the other actually starts filling. 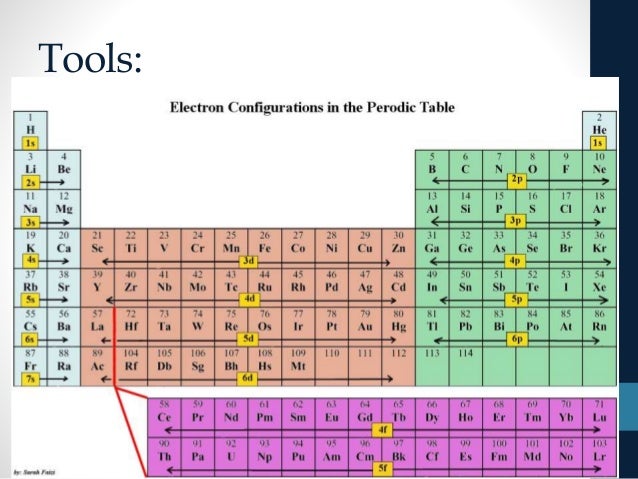 
The actual filling order is more complicated. This pattern will give you the correct configuration for all but about 19 elements. There are no known elements that, in their ground state, have electrons in a subshell beyond 7p. What you do is you start assigning electrons to the subshells using the following pattern (you completely fill up one subshell before moving onto the next higher) and you keep going until the total number of electrons assigned is equal to the atomic number:ġs, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p 
However, in the simplified version of these rules that is understandable to a general chemistry student, there are about 19 elements that are exceptions. Given the atomic number, there is a set of rules that allow you to determine the electron configuration. Without that, you cannot determine the electron configuration. \), and \(Cu^ \).įor example, you would expect the electron configuration of Cu to be: 1s 22s 22p 63s 23p 64s 23d 9 (paramagnetic, 1 unpaired electron) and when it loses one electron to form the Cu with the following electron configuration: 1s 22s 22p 63s 23p 64s 23d 8(paramagnetic 2 unpaired electrons).You must know the atomic number of the element. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
